(Enforcement and Intelligence cell)
FDA Bhawan, Kotla Road
New Delhi-110002
Date: 28 AUG 2025
NOTICE
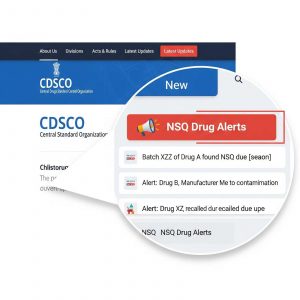
Subject: Availability of Not of Standard Quality (NSQ) Alert on New Link in CDSCO Website-reg.
- The NSQ alerts are published on a monthly basis by CDSCO and are made available on the CDSCO website.
- In this regard, CDSCO has taken new initiative and the NSQ alerts can now be accessed via a new dedicated link on the official CDSCO website.
- In the newly implemented NSQ alerts, details like Name of product, manufacturing details, NSQ results etc., are easily searchable.
All stakeholders can visit the CDSCO website and follow the updated link for the latest NSQ alerts and related information.
Yours faithfully,
(Signed)
(Dr. Rajeev Singh Raghuvanshi)
Drugs Controller General (India)

To
- All Stakeholders
- CDSCO website
(Gazette Notification – Draft Rules)
MINISTRY OF HEALTH AND FAMILY WELFARE
(Department of Health and Family Welfare)
NOTIFICATION
New Delhi, the 27th August, 2025
G.S.R. 587(E). — The following draft of certain rules further to amend the New Drugs and Clinical Trials Rules, 2019, which the Central Government proposes to make, in exercise of the powers conferred by sub-section (1) of section 12 and sub-section (1) of section 33 of the Drugs and Cosmetics Act, 1940 (23 of 1940), after consultation with the Drugs Technical Advisory Board is hereby published for information of all persons likely to be affected thereby, and notice is hereby given that the said draft rules shall be taken into consideration after the expiry of a period of thirty days from the date on which the copies of the Gazette of India containing these draft rules are made available to the public.
Objections and suggestions which may be received from any person within the period specified above will be considered by the Central Government.
Objections and suggestions, if any, may be addressed to the Under Secretary (Drugs), Ministry of Health and Family Welfare, Government of India, Room No. 545, A Wing, Nirman Bhavan, New Delhi – 110011 or emailed at drugs-div-mohfw@gov.in
DRAFT RULES
(i) These rules may be called the New Drugs and Clinical Trials (….. Amendment) Rules, 2025
(ii) These rules shall come into force from the date as specified by the Government at the time of final publication of the rules in the Official Gazette.
In the New Drugs and Clinical Trials Rules, 2019, after sub-rule (2) of rule 31, the following proviso shall be inserted, namely: —
“Provided that in case of single-dose, two-period, two-sequence, two-treatment, bioavailability or bioequivalence studies in normal healthy adult human volunteers (for export purpose only), of oral dosage form of a drug (other than drugs of Cytotoxic, Hormone, Narcotic and Psychotropic substances categories and not a drug of Narrow Therapeutic Index) and having highly variable pharmacokinetic parameters already approved in the Country or any one of the countries namely, USA, EU, Japan, Australia, Canada and UK, the studies may be conducted after submission of an online application and acknowledgement to the Central Licensing Authority, subject to the following conditions:
- The application for the notification shall be accompanied with approval of the ethics committee registered with the Central Licensing Authority.
- The Ethics Committee shall maintain separately the record of review and approval of such bioavailability or bioequivalence study. The record of such notification along with its acknowledgement shall be reviewed by the Central Licensing Authority at the time of renewal of the Registration of the Ethics Committee.
- The sample size should not be more than 48.”
- In the New Drugs and Clinical Trials Rules, 2019, after sub-rule (1) of rule 33, the following proviso shall be inserted, namely: —
“Provided that in case of bioavailability or bioequivalence studies as referred in proviso to sub-rule (2) of rule 31, the bioavailability or bioequivalence studies may be conducted by submitting an online application in Form CT-45 as notification.”
- In the New Drugs and Clinical Trials Rules, 2019, after sub-rule (2) of rule 33 shall be substituted, namely:
An application for grant of permission to conduct bioavailability or bioequivalence study of any new drug or investigational new drug, or for notification for initiation of bioavailability or bioequivalence studies as referred in proviso to sub-rule (2) of rule 31, as the case may be, shall be accompanied by a fee as specified in Sixth Schedule and such other information and documents as specified in the Fourth Schedule.”
PROVIDED that no fee shall be payable for conducting a bioavailability or bioequivalence study by an institution or organisation owned or funded wholly and partially by the Central Government or a State Government.
[F. No. X.11014/06/2025-DR]
NIKHIL GAUR, Jt. Secy.
